40 vs 60 L/min: What’s the Difference Between Vapotherm® High Velocity Therapy and High Flow Nasal Cannula?
Vapotherm’s high velocity therapy is a tool for treating respiratory distress. Vapotherm does not practice medicine or provide medical services or advice. Any guidelines provided herein are based on an assessment of peer-reviewed published literature, physician interviews, and physiological modeling. Practitioners should refer to the full indications for use and operating instructions of any products referenced before use.
Vapotherm high velocity therapy often gets conflated or confused with commodity high flow oxygen products, also commonly known as High Flow Nasal Cannula (HFNC). Many studies don’t differentiate between the two, though this is slowly changing in the medical field as randomized controlled trial evidence showed high velocity therapy to have outcomes comparable to Non-Invasive Positive Pressure Ventilation (NiPPV) when treating adult emergency department patients in undifferentiated respiratory distress.[1] It’s also slowly changing with the FDA’s creation of a new product code for the Precision Flow Hi-VNI™, placing the technology into a different category from commodity high flow altogether.
However, many practitioners are still wondering what exactly the difference is. The easiest way to describe it is that high velocity therapy is Mask-Free NIV®, and offers ventilatory support for patients in respiratory distress, including hypercapnia, hypoxemia, dyspnea, respiratory distress secondary to other medical conditions. Meanwhile commodity high flow oxygen products provide oxygenation support. There are many design differences that may lead to the outcome differences between the modalities.
Why Does Vapotherm High Velocity Therapy Go Up to 40 L/min? — Assumptions About Flow Rates and Why More Is Not Always Better
High velocity therapy and commodity high flow oxygen products have similar goals when it comes to maintaining patient comfort and tolerance while delivering respiratory support. One of the easily spotted differences between the two is the liter flow maximum. However, there is an important, less visible difference in the velocity at which flow is delivered. Understanding the mechanisms behind these differences can also help shed light on the differences in patient outcomes.
If the two modalities had identical characteristics when delivering flow, one would assume that a higher maximum liter flow would be more effective. For one, it could allow clinicians a greater range of titration depending on the patient’s respiratory needs. A second frequently made assumption is that having higher flow rates could potentially rescue patients presenting with moderate or even severe respiratory distress by more effectively maintaining oxygenation and supporting ventilation via the greater flow volume. However, high velocity therapy and commodity high flow oxygen products don’t deliver flow the same way. There are significant differences that impact patient outcomes.
The Difference Between Flow in high velocity vs HFNC
The five commonly accepted mechanisms of action for HFNC in general are:[2]
- Washout of upper airway dead space
- Reduction of work of breathing (WOB) through provision of adequate flow
- Improvement of lung mechanics through adequately humidified and warmed gas
- Reduction of energy cost for conditioning (heating and humidifying) gas
- Provision of distending pressure (akin to CPAP)
While there are differences in the ways high velocity therapy achieves optimally conditioned gas, the first two mechanisms of action — washout of dead space and reduction of WOB — are the most relevant for this discussion on flow rates.
High velocity therapy and dead space flushing: High velocity therapy includes an important velocity-based approach to flushing the nasopharyngeal dead space. Unlike most commodity high flow oxygen products, high velocity therapy uses small-bore cannulas. In the 2016 study “Computational Fluid Dynamics Modeling of Extrathoracic Airway Flush: Evaluation of High Flow Nasal Cannula Design Elements” Miller and colleagues demonstrated that small-bore cannula prongs flush the dead space faster than large-bore cannulas (2.2 seconds for small-bore cannula vs. 3.6 seconds for large-bore cannula).[3] This is clinically meaningful because with the velocity the small-bore cannulas generate, a lower flow rate is needed to achieve an effective, rapid flush. Figure 1 illustrates how high velocity therapy (labeled as high velocity nasal insufflation [HVNI]) compares to commodity high flow oxygen products when generating velocity. high velocity therapy is clinically effective below 40 L/min.
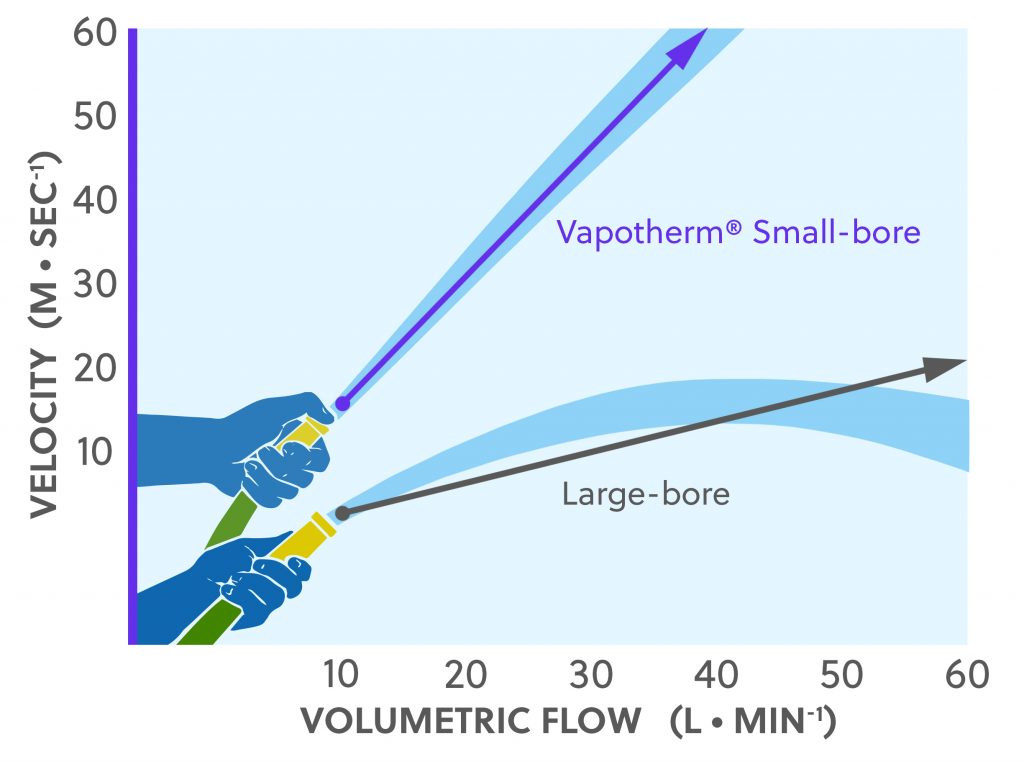
High velocity therapy and WOB reduction: The fact that small-bore cannulas reduce the time required to fully purge the upper airway dead space[3] is significant because as the respiratory rate of a patient in respiratory distress increases, the time between breaths decreases. By quickly clearing the upper airway dead space of end-expiratory gas rich in CO2, high velocity therapy helps patients breathe directly from a fresh gas reservoir and thereby reduces their WOB.
One limitation of small-bore cannulas is that they generate a significant amount of back-pressure. However, Vapotherm’s Precision Flow capital unit was designed to work specifically with small-prong cannulas and can therefore effectively deliver high velocity therapy.
Commodity High Flow Oxygen Products: Conventional HFNC also flush the nasopharyngeal dead space in order to reduce a patient’s WOB. As previously noted, most of those systems have a large-bore cannula design and/or are not designed to effectively withstand the back pressure generated with high flows through a small-bore cannula. Therefore, the key difference is that the mechanism of action involved in these systems relies on much higher flow rates — such as 60 L/min — that try to compensate for lack of velocity through volumetric flush.
If It’s the Same Volume, Why Does It Matter When It’s Delivered Faster?
It is sometimes hard to easily visualize why velocity would matter. After all, if one device delivers 25 liters of flow per minute and another device does too, doesn’t the patient receive 25 liters of flow per minute? The answer is yes, but it matters just how the flow is delivered.
A good analogy is a garden hose. If you have two garden hoses running side by side delivering the exact same volume of flow, their performance would be equivalent. However, as soon as you place your thumb over the spigot of one, thereby occluding part of the opening (akin to a small-bore cannula), the water would spray farther. The volume would remain the same, but the water coming through the smaller opening would flow faster, i.e. at a greater velocity, as depicted in Figure 2.
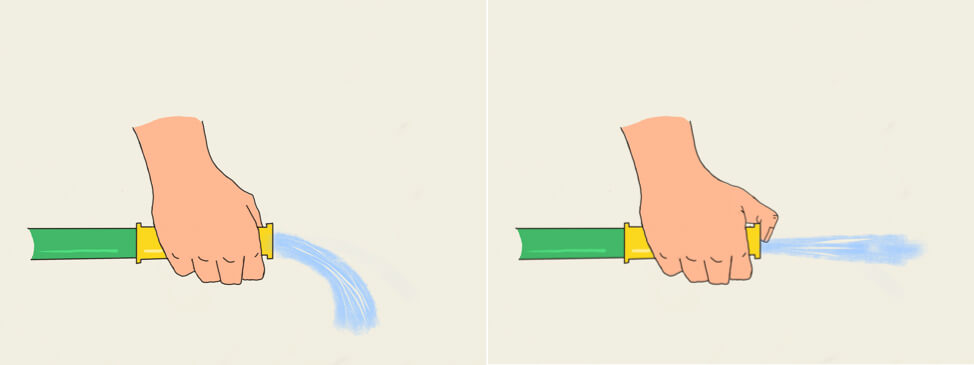
The greater velocity generates a greater turbulent energy that in turn ensures a faster flushing of CO2-rich end-expiratory gas in the upper airway dead space.[2]
The Difference Between Clinical Outcomes in High Velocity Therapy vs HFNC
At the time of this writing Vapotherm’s high velocity therapy is the only Mask-Free NIV that includes a velocity-based mechanism of action which gives it clinical efficacy comparable to Non-Invasive Positive Pressure Ventilation (NiPPV).[1] In 2018, The Annals of Emergency Medicine published a multi-center randomized, controlled, noninferiority trial by Doshi and colleagues, comparing high velocity therapy to Non-Invasive Positive Pressure Ventilation, titled “High Velocity Nasal Insufflation in the Treatment of Respiratory Failure”. The trial found high velocity therapy to be non-inferior to NiPPV in regard to risk for intubations in adults presenting in the ED with undifferentiated respiratory distress, including hypercapnia. In other words, there was no increased risk for intubations.
There is evidence demonstrating comparable outcomes to NCPAP and bi-level PAP in the neonatal population as well. Lavizzari and colleagues published the results of a randomized, monocentric noninferiority trial in August 2016, titled “Heated, Humidified High-Flow Nasal Cannula vs Nasal Continuous Positive Airway Pressure for Respiratory Distress Syndrome of Prematurity A Randomized Clinical Noninferiority Trial” in JAMA Pediatrics. Although not explicitly investigating high velocity therapy, this trial was exclusively conducted on Vapotherm’s high velocity therapy. The primary outcomes found that HFNC (in this case Vapotherm high velocity therapy) is noninferior to NCPAP or bi-level PAP for the purpose of primary support of infants with RDS.[4]
By comparison, studies that examine how commodity high flow oxygen products compare to noninvasive ventilation (NIV) tend to focus only on patients with symptoms of hypoxemia and exclude hypercapnia. For example, in the multicenter randomized controlled trial published in 2017, Frat and colleagues concluded that “HFNC seems to be a good alternative to standard oxygen and NIV as treatment for patients with hypoxemic ARF.”[5] Similarly, a randomized clinical trial published in 2016 by Hernandez and colleagues investigated the question: “Is high-flow nasal cannula noninferior to noninvasive ventilation for preventing reintubation and post extubation respiratory failure?”[6] They found noninferiority for HFNC to NIV for preventing reintubation in adult patients. However, as with Frat et al, their study design once again excluded hypercapnic patients.
In short, there is evidence that demonstrates that high velocity therapy is a tool for treating undifferentiated respiratory distress, including hypercapnia. There is no evidence to demonstrate this for conventional HFNC devices.
Clinical Efficacy as the Deciding Factor in Flow Rate
While clinicians should titrate flow to clinical effect, Vapotherm high velocity therapy generally achieves clinical efficacy between 25-35 L/min in adults and 6-8 L/min in neonates and for pediatric patients it is recommended to set the flow at 1.5-2 L/min per kilogram of bodyweight, up to 8kg[7].
As a point of reference, the starting flow rates for respiratory distress in all-comers in the ED from Doshi and colleagues’ randomized controlled trial were 35 L/min.[1] The average flow rate across the trial was 30 L/min.
Ultimately, when it comes to deciding which modality and which flow rates to use, it is important for clinicians to follow evidence-based practice.
Learn more about Vapotherm high velocity therapy
REFERENCES
[1] Doshi, Pratik et al. High-Velocity Nasal Insufflation in the Treatment of Respiratory Failure: A Randomized Clinical Trial. Annals of Emergency Medicine, 2018. Published online ahead of print. https://www.ncbi.nlm.nih.gov/pubmed/29310868
[2] Dysart K, Miller T, Wolfson M, Shaffer T. Research in high flow therapy: Mechanisms of action. Respiratory Medicine. 2009; 103: 1400-05. (Review)
[3] Miller TL, Saberi B, Saberi S (2016) Computational Fluid Dynamics Modeling of Extrathoracic Airway Flush: Evaluation of High Flow Nasal Cannula Design Elements. J Pulm Respir Med 6:376. doi: 10.4172/2161-105X.1000376. (Bench, Prospective, Not Randomized) https://www.omicsonline.org/open-access/computational-fluid-dynamics-modeling-of-extrathoracic-airway-flush-evaluation-of-high-flow-nasal-cannula-design-elements-2161-105X-1000376.php?aid=81462
[4] Lavizzari A, Colnaghi M, Ciuffini F, Veneroni C, Musumeci S, Cortinovis I, Mosca F. “Heated, humidified high-flow nasal cannula vs nasal continuous positive airway pressure for respiratory distress syndrome of prematurity – a randomized clinical noninferiority trial.” JAMA Pediatr. 2016 Aug
[5] Frat, Jean-Pierre, Rémi Coudroy, Nicolas Marjanovic, and Arnaud W. Thille. “High-flow nasal oxygen therapy and noninvasive ventilation in the management of acute hypoxemic respiratory failure.” Ann Transl Med. 2017 Jul; 5(14): 297. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5537116/
[6] Hernández, Gonzalo, Concepción Vaquero, Laura Colinas, et al. “Effect of Postextubation High-Flow Nasal Cannula vs Noninvasive Ventilation on Reintubation and Postextubation Respiratory Failure in High-Risk Patients A Randomized Clinical Trial.” JAMA. 2016;316(15):1565-1574. doi:10.1001/jama.2016.14194 https://jamanetwork.com/journals/jama/fullarticle/2565304
[7] Weiler, Thomas MD, Asavari Kamerkar DO, Justin Hotz RRT, Patrick A.Ross MD, Christopher J.L.Newth MD, FRCPC, Robinder G.Khemani MD, MsCI. The Relationship between High Flow Nasal Cannula Flow Rate and Effort of Breathing in Children. The Journal of Pediatrics Volume 189, October 2017, Pages 66-71.e3. https://doi.org/10.1016/j.jpeds.2017.06.006