How to Select Clinically Effective Liter Flows for Hypercapnic Patients?

Vapotherm’s high velocity therapy can be used to treat respiratory distress in hospital settings. Vapotherm does not practice medicine or provide medical services. These guidelines are based on an assessment of peer-reviewed published literature, physician interviews, and physiological modeling. Providers should refer to the full indications for use, operating instructions, and prescribing information of any products referenced herein before prescribing them.
Although High Flow Nasal Cannula (HFNC) has been demonstrated to be non-inferior to noninvasive ventilation (NIV) for treating hypoxemia, the trials which arrived at those results excluded hypercapnic patients.[1,2] Evidence like this is in part why clinicians routinely turn to noninvasive positive pressure ventilation (NIPPV) when treating hypercapnic patients.However, Vapotherm’s® high velocity therapy has a nasal cannula interface, but has been found to have similar efficacy as NIPPV in the treatment of undifferentiated respiratory distress in adults. High velocity therapy is the only mask-free respiratory support alternative for the treatment of undifferentiated respiratory distress — including for treatment of hypercapnia. There are substantial design differences between high velocity therapy and conventional HFNC which may account for this difference in outcomes. The below graph illustrates how high velocity therapy compares to NIPPV when it comes to reducing hypercapnia in Emergency Department adult all-comers during a recent multi-center randomized trial by Doshi and colleagues.[3]
High velocity therapy efficiently reduces CO2
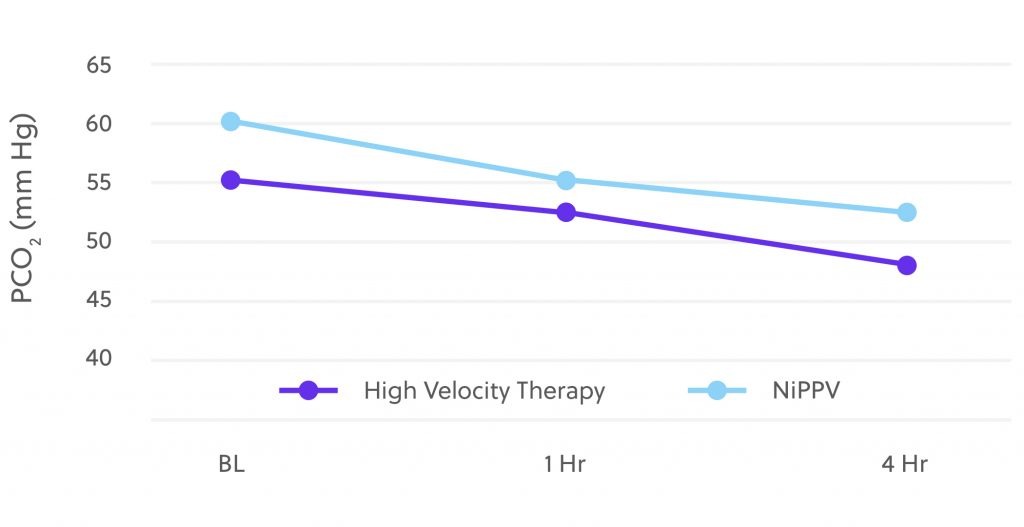
The data shows there are no significant differences in the CO2 reduction over time between Vapotherm’s high velocity therapy and gold standard NIPPV.
Selecting Liter Flows
While clinicians should titrate flow to clinical effect, Vapotherm high velocity therapy generally achieves clinical efficacy between 25-35 LPM in adults, 6-8 LPM[4,5,6] in neonates, and for pediatric patients it is recommended to set the flow at 1.5-2LPM per kilogram of bodyweight, up to 8kg[7]. You can also consult the pediatric flow calculator to compute recommended starting flows for larger pediatric patients.As a point of reference, the starting flow rates for respiratory distress in Doshi and colleagues’ randomized controlled trial in adults were 35 LPM.[3] The average flow rate across the trial was 30 LPM.
Learn more about Vapotherm high velocity therapy
REFERENCES
[1] Frat, Jean-Pierre, Rémi Coudroy, Nicolas Marjanovic, and Arnaud W. Thille. High-flow nasal oxygen therapy and noninvasive ventilation in the management of acute hypoxemic respiratory failure. Ann Transl Med. 2017 Jul; 5(14): 297. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5537116/
[2] Hernández, Gonzalo, Concepción Vaquero, Laura Colinas, et al. Effect of Postextubation High-Flow Nasal Cannula vs Noninvasive Ventilation on Reintubation and Postextubation Respiratory Failure in High-Risk Patients A Randomized Clinical Trial. JAMA. 2016;316(15):1565-1574. doi:10.1001/jama.2016.14194 https://jamanetwork.com/journals/jama/fullarticle/2565304
[3] Doshi P, Whittle JS, Bublewicz M, et al. High-Velocity Nasal Insufflation in the Treatment of Respiratory Failure: A Randomized Clinical Trial. Ann Emerg Med 2018;72:73-83 e5. https://www.annemergmed.com/article/S0196-0644(17)31968-6/abstract
[4] Yoder BA, B Manley, C Collins, K Ives, A Kugelman, A Lavizzari, and M McQueen. “Consensus approach to nasal high-flow therapy in neonates.” Journal of Perinatology (2017) 00, 1–5.
[5] Lavizarri A, Colnaghi M, Ciuffini F, Veneroni C, Musumeci S, Cortinovis I, Mosca F. “Heated, humidified high-flow nasal cannula vs nasal continuous positive airway pressure for respiratory distress syndrome of prematurity – a randomized clinical noninferiority trial.” JAMA Pediatr. 2016 Aug 8.
[6] Kugelman A, Riskin A, Said W, Shoris I, Mor F, et al. (2015) A randomized pilot study comparing heated humidified high-flow nasal cannulae with NIPPV for RDS. Pediatr Pulmonol 50(6): 576-583.
[7] Weiler, Thomas MD, Asavari Kamerkar DO, Justin Hotz RRT, Patrick A.Ross MD, Christopher J.L.Newth MD, FRCPC, Robinder G.Khemani MD, MsCI. The Relationship between High Flow Nasal Cannula Flow Rate and Effort of Breathing in Children. The Journal of Pediatrics Volume 189, October 2017, Pages 66-71.e3. https://doi.org/10.1016/j.jpeds.2017.06.006